Aqueous Stability of Zirconium Clusters, Including the Zr(IV) Hexanuclear Hydrolysis Complex [Zr6O4(OH)4(H2O)24]12+, from Density Functional Theory
Abstract
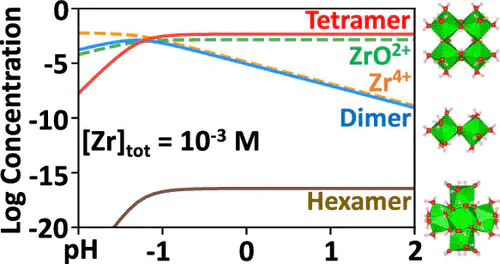
Framework materials constitute a broad family of solids that range from zeolites and metal–organic frameworks (MOFs) to coordination polymers. The synthesis of such network structures typically rely on precursor molecular building blocks. As an example, the UiO-66 MOF series is constructed of hexanuclear [Zr6O4(OH)4(CO2)12] cluster nodes and linear carboxylate linkers. Unfortunately, these Zr MOF cluster nodes cannot currently be manufactured in a sustainable way, motivating a search for “green” alternative synthesis methods. Stabilizing the hexanuclear Zr(IV) cluster (i.e., the hexamer, {Zr612+}) without the use of organic ligation would enable the use of environmentally friendly solvents such as water. The Zr(IV) tetranuclear cluster (i.e., the tetramer, {Zr48+}) can be stabilized in solution with or without organic ligands, yet the hexamer has yet to be synthesized without supporting ligands. The reasons why certain zirconium clusters are favored in aqueous solution over others are not well understood. This study reports the relative thermodynamic instability of the hypothetical hexamer {Zr612+} compared to the ubiquitous {Zr48+} tetramer. Density functional theory calculations were performed to obtain the hydrolysis Gibbs free energy of these species and used to construct Zr Pourbaix diagrams that illustrate the effects of electrochemical potential, pH, and Zr(IV) concentration. It was found that the aqueous {Zr612+} hexamer is ∼17.8 kcal/mol less stable than the aqueous {Zr48+} tetramer at pH = 0, V = 0, and [Zr(IV)] = 1 M, which is an energy difference on the order of counterion interactions. Electronic structure analyses were used to explore trends in the highest occupied molecular orbital–lowest unoccupied molecular orbital gap, frontier molecular orbitals, and electrostatic potential distribution of these clusters. The evidence suggests that the aqueous {Zr612+} hexamer may be promoted with more strategic syntheses incorporating minimal ligands and counterions.